
May 29, 2025
Innovation Fund Spotlight: State-of-the-Art Genome Editing To Understand Disease and Develop New Therapies
Read Article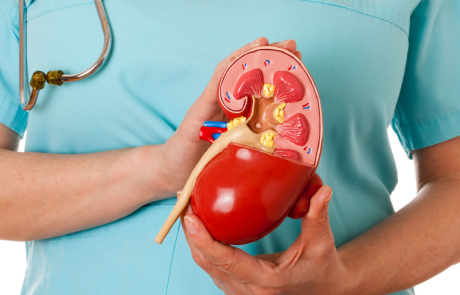
May 7, 2025
Finding the Root Cause of Lupus
For more than 20 years, Steven Ziegler, PhD, has wondered if an altered version of a protein might play a key role in autoimmune diseases. Now, he finally has the tools to investigate further – and his work could lead to key insights about how and why lupus happens.
May 7, 2025
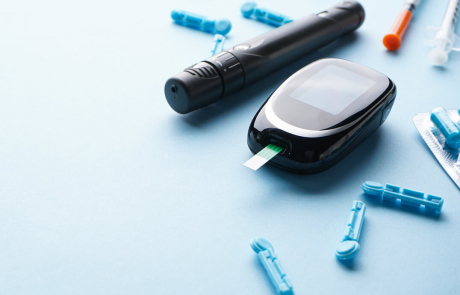
Autoantibody Reversion: A Mystery That Could Help Prevent T1D
At first, Cate Speake, PhD, and her team weren’t sure that a mysterious phenomenon called autoantibody reversion was actually happening. But no matter how they sliced the data, they saw the same pattern. Learn how autoantibody reversion could unlock new clues to preventing type 1 diabetes.
May 7, 2025
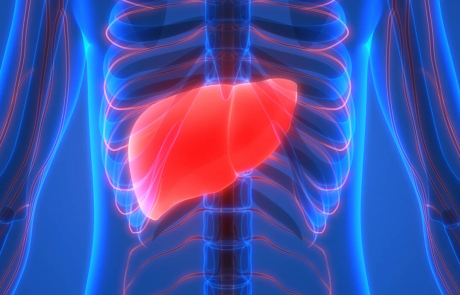
A New Approach to Autoimmune Disease Treatment Takes a Big Step Forward
BRI’s Buckner Lab has been pioneering an innovative approach that turns cells that cause T1D into cells that protect you from T1D. Now, new research from Soo Jung Yang, PhD, and Ritika Tewari, PhD, shows this strategy may help treat other autoimmune diseases too.
February 10, 2025
Innovation Grant Yields Exciting Advances and Additional Funding
Learn how lab-grown human beta cells are helping Eddie James, PhD, and other BRI researchers answer important questions in type 1 diabetes.

February 10, 2025
Computational Skills Grant Fuels Collaboration and Discovery
Learn how Susana Orozco, PhD, Hannah DeBerg, PhD, and Jessica Hamerman, PhD, are working together to apply the latest tools to long-standing research questions.
February 10, 2025
Catching Autoimmune Diseases Before They Start
Find out about new advances in autoimmune disease screening from a team led by Cate Speake, PhD.

February 10, 2025
A Letter from our President
Read exciting updates and learn what’s on the horizon for 2025 from BRI President Jane Buckner, MD.

January 28, 2025
Interview with Dr. Megan Smithmyer: Type 1 Diabetes Biorepository (BRIDge Study)
Dr. Smithmyer is a staff scientist at Benaroya Research Institute (BRI) in the Center for Interventional Immunology. She has kindly agreed to speak with us about what the BRIDge Biorepository is and its importance to the work that scientists like her do in the realm of type 1 diabetes.

October 23, 2024
Innovation Fund Spotlight: Single-Cell CRISPR Screening To Explore Genetics and Autoimmune Diseases
Together, Jessica Hamerman, PhD, and John Ray, PhD, are exploring the genetic roots of autoimmune disease using single-cell CRISPR screening.

October 23, 2024
Innovation Fund Spotlight: Lab-Grown Beta Cells Meet State-Of-The-Art Imaging To Answer New Questions About Type 1 Diabetes
BRI investigators Eddie James, PhD, and Caroline Stefani, PhD, are teaming up to take a unique approach to type 1 diabetes research.


